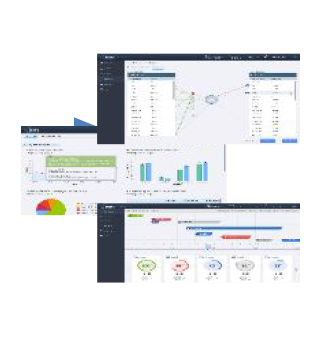
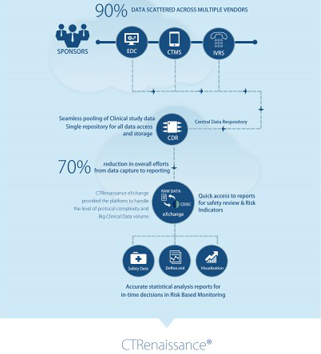
MaxisIT® is the premier, worldwide contributor in improving ways of leveraging data and information and speeding up decision-making process in the conduct of clinical research and development.
 Menu
≡
╳
Menu
≡
╳
-
WHAT WE DO
- CLINICAL SOLUTIONS
- SERVICES
-
WHO WE ARE
-
RESOURCES
- CAREERS
- CONTACT US