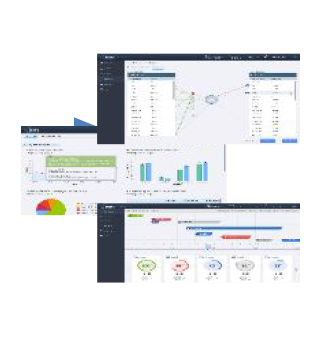
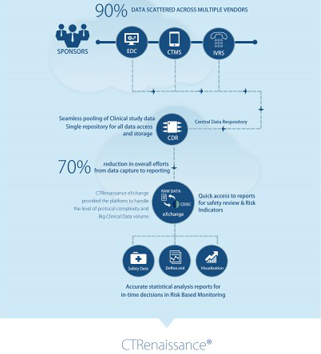
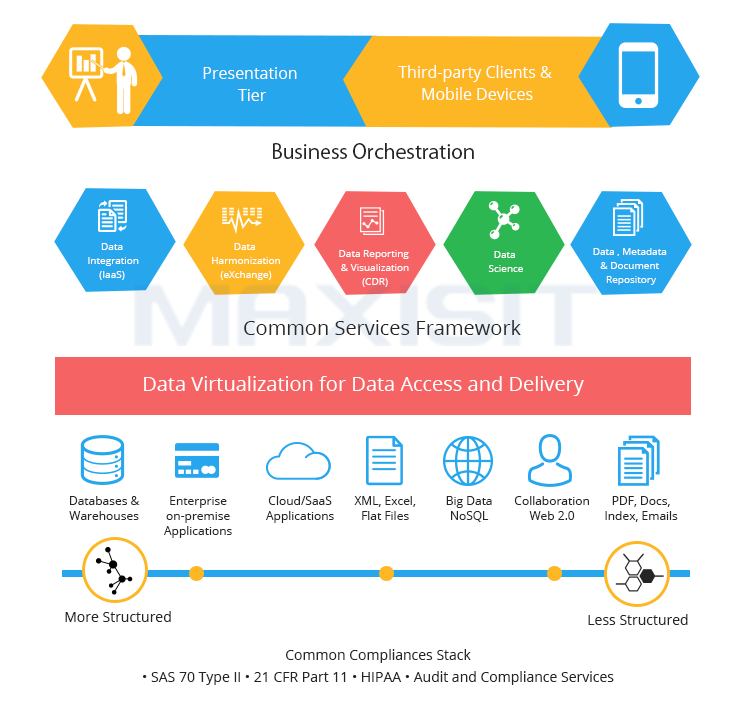
Get a pre-validated, metadata driven, self-service solution which is configured to tailor-meet your business needs. It offers self-service solutions for data integration, extraction, cleansing, standardization, validation, data management and reporting and analysis of regulatory compliant setting. Get a ubiquitous Data Science Workbench with an option to perform exploratory analysis.
 Menu
≡
╳
Menu
≡
╳
-
WHAT WE DO
- CLINICAL SOLUTIONS
- SERVICES
-
WHO WE ARE
-
RESOURCES
- CAREERS
- CONTACT US